Oxygen is a colorless, tasteless, odorless gas that is slightly heavier than air.
It is nonflammable but will support combustion with other elements.
In its free state, oxygen is one of the most common elements.
The atmosphere is made up of approximately 21 parts of oxygen and 78 parts of nitrogen, the remainder being rare gases.
Rusting of ferrous metals, discoloration of copper, and the corrosion of aluminum are all due to the action of atmospheric oxygen, known as oxidation.
For welding, oxygen cylinders are measured in cubic feet and come in 3 sizes (80, 122 and 244). The cylinders are pressurized to 2,220 psi (pounds per square inch). The cylinders fittings for oxygen are right-handed (clockwise for tightening).
Every cylinder includes a safety device to release pressure in high temperatures that might cause a dangerous condition.
Pure oxygen does not explode or burn. It is an accelerator that causes other gases to burn hotter, faster and at lower temperatures.
CAUTION
Always refer to oxygen as oxygen, never as air.
Combustibles should be kept away from oxygen, including the cylinder, valves, regulators, and other hose apparatus. Oxygen cylinders and apparatus should not be handled with oily hands or oily gloves. Pure oxygen will support and accelerate combustion of almost any material, and is especially dangerous in the presence of oil and grease. Oil and grease in the presence of oxygen may spontaneously ignite and burn violently or explode. Oxygen should never be used in any air tools or for any of the purposes for which compressed air is normally used.
Oxygen is heavier than air (1.105). If an oxygen cylinder is leaking, the oxygen will settle into low areas. If the concentration of oxygen is 23.5% or higher, it will cause a gas to explode or burn 8 times faster.
Production of Bottled Oxygen
Bottle oxygen is obtained commercially either by the liquid air process or by the electrolytic process.
Liquid Air Process:
In the liquid air process, air is compressed and cooled to a point where the gases become liquid. As the temperature of the liquid air rises, nitrogen in a gaseous form is given off first, since its boiling point is lower than that of liquid oxygen. These gases, having been separated, are then further purified and compressed into cylinders for use. The liquid air process is by far the most widely used to produce oxygen.
Electrolyte Process:
In the electrolytic process, water is broken down into hydrogen and oxygen by the passage of an electric current. The oxygen collects at the positive terminal and the hydrogen at the negative terminal. Each gas is collected and compressed into cylinders for use.
Oxygen Cylinder
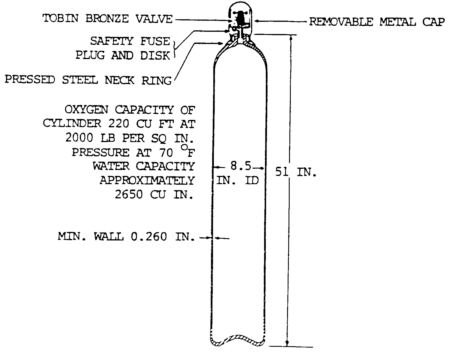
A typical bottle oxygen cylinder is shown in figure 5-7 below.
It is made of steel and has a capacity of 220 cu ft at a pressure of 2000 psi (13,790 kPa) and a temperature of 70°F (21°C).
Attached equipment provided by the oxygen supplier consists of an outlet valve, a removable metal cap for the protection of the valve, and a low melting point safety fuse plug and disk.
The cylinder is fabricated from a single plate of high-grade steel so that it will have no seams and is heat-treated to achieve maximum strength.
Because of their high pressure, oxygen cylinders undergo extensive testing prior to their release for work, and must be periodically tested thereafter.
Related: Welding Gas Types
Welding Oxygen Basics

A cylinder truck will keep cylinders from being knocked over when used.
- More than 2000 psi when full.
- The opening at the top of the bottle is smaller than a pencil, if damaged, the bottle can become a projectile and could cause injury or property damage.
- Never allow a tank to go empty as this may allow one gas to backflow into the other gas line or bottle.
- Keep fittings, regulators and hoses free from oil, dirt and gas as they can explode under high-pressure oxygen.
- Bottles must always be chained.
- The bottle must be capped any time a regulator is not attached to the valve.
- New clam style safety caps allow bottles to be capped even when a regulator is attached.
- Oxygen bottles should never be stored with flammables/combustibles.
- When stored must have a 5’ min. height wall with a 30 minute fire rating (usually cinder block)
- The correct way to move a bottle is chained in a 2 wheel dolly, not by rolling with your hands. The bottle could fall and cause injury.
- Always store bottle oxygen separately from other fuel-gas cylinders (a minimum distance of 20 feet) or use a noncombustible barrier at least 5 feet away with a fire resistance rating of at least 1/2 hour.
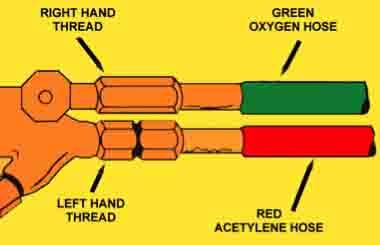
Reverse Flow Danger
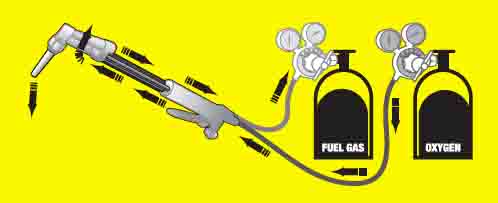
Reverse flow occurs when the bottle oxygen cylinder is near empty at the same time another gas, such as acetylene is not. The result is that the fuel gas such as acetylene can flow into the oxygen hose and regulator.
If this occurs, an explosion can occur if the gases are lighted before bleeding off the oxygen.
To prevent this problem never use a bottle of oxygen cylinder that is almost empty. Empty is considered any cylinder that is 50 psi or lower.
Operators should always independently bleed hoses before lighting a welding torch.
Never light both gases at once (exception is the universal pressure torch which is designed to prevent the risk of reverse flow).
Backfire and Flashback Danger
A backfire occurs when the torch flame burns back into the torch tip and is extinguished with a loud popping sound. It can be caused by the tip of the torch touching the work, or if there is not enough gas pressure.
Frequent backfires can damage a welding torch.
A flashback occurs when the flame front does not extinguish but burns back beyond the mixing chamber through the hoses and into the regulator and gas supply. The size of a potential explosion should this occur is the equivalent of 20 sticks of dynamite.
To help avoid problems make sure that the internal check valve is working properly. Flashback arrestors will stop flashbacks.
If flashback occurs immediately turn off the oxygen, then the acetylene and allow all units to cool.