What is metal brazing?
Brazing is a group of welding processes which produces coalescence of materials by heating to a suitable temperature and using a filler metal having a liquidus above 840ºF (449ºC) and below the solidus of the base metals.
The filler metal is distributed between the closely fitted surfaces of the joint by capillary attraction.
Brazing is distinguished from soldering in that soldering employs a filler metal having a liquidus below 840ºF (449ºC).
When brazing with silver alloy filler metals (silver soldering), the alloys have liquidus temperatures above 840ºF (449ºC).
Brazing is only used on ferrous metals because the solder melts at 960 degrees Celcius, a point above the melting point of non-ferrous metals.
Brazing must meet each of three criteria:
- The parts must be joined without melting the base metals.
- The filler metal must have a liquidus temperature above 840ºF (449ºC).
- The filler metal must wet the base metal surfaces and be drawn onto or held in the joint by capillary attraction.
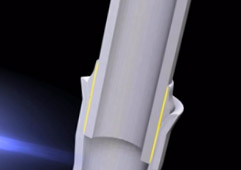
Differences Between Brazing vs Welding and Soldering
Brazing is not the same as braze welding.
Brazing is capillary, with heat drawing a filler metal using capillary action. Capillary action refers to the way filler metal is drawn into a properly fitted joint. A simple example is the way water adheres to and is drawn up a straw in opposition to external forces such as gravity.
Braze welding bridges a gap using filler metal that is melted and deposited in fillets and grooves exactly at the points it is to be used. Limited base metal fusion may occur in braze welding.
Soldering is a process that joins materials with a filler metal that has a liquidus not greater than 842ºF (450ºC). Like brazing, the filler metal moves into the joint with a capillary action.
To achieve a good joint using any of the various brazing processes, the parts must be properly cleaned and protected by either flux or the atmosphere during heating to prevent excessive oxidation.
The parts must provide a capillary for the filler metal when properly aligned, and a heating process must be selected that will provide proper brazing temperatures and heat distribution.
Principles
Capillary flow is the most important physical principle which ensures good brazements providing both adjoining surfaces molten filler metal.
The joint must also be properly spaced to permit efficient capillary action and resulting coalescence. More specifically, capillarity is a result of surface tension between base metal(s), filler metal, flux or atmosphere, and the contact angle between base and filler metals.
In actual practice, brazing filler metal flow characteristics are also influenced by considerations involving fluidity, viscosity, vapor pressure, gravity, and by the effects of any metallurgical reactions between the filler and base metals.
The brazed joint, in general, is one of a relatively large area and very small thickness. In the simplest application of the process, the surfaces to be joined are cleaned to remove contaminants and oxide. Next, they are coated with flux or a material capable of dissolving solid metal oxides present and preventing new oxidation. The joint area is then heated until the flux melts and cleans the base metals, which are protected against further oxidation by the liquid flux layer.
Brazing filler metal is then melted at some point on the surface of the joint area. Capillary attraction is much higher between the base and filler metals than that between the base metal and flux. Therefore, the flux is removed by the filler metal. The joint, upon cooling to room temperature, will be filled with solid filler metal. The solid flux will be found on the joint surface.
High fluidity is a desirable characteristic of brazing filler metal because capillary attraction may be insufficient to cause a viscous filler metal to run into tight-fitting joints.
Brazing is sometimes done with active gas, such as hydrogen, or in an inert gas or vacuum. Atmosphere brazing eliminates the necessity for post-cleaning and ensures absence of corrosive mineral flux residue. Carbon steels, stainless steels, and superalloy components are widely processed in atmospheres of reacted gases, dry hydrogen, dissociated ammonia, argon, and vacuum. Large vacuum furnaces are used to braze zirconium, titanium, stainless steels, and refractory metals. With good processing procedures, aluminum alloys can also be vacuum furnace brazed with excellent results.
Brazing is a process preferred for making high strength metallurgical bonds and preserving needed base metal properties because it is economical.
Smooth Joints Created Using Fillet Brazing over Acetylene Welding for Bicycle Frame
Tips
1: Allow for Correct Joint Clearance
The two metals being joined need to have a close and optimal fit in order for the capillary action of the brazing alloy filler metal to be drawn in. An engineer will anticipate the right amount of fit in order to maximize strength per the table below.
Recommended brazed joint clearance at brazing temperatures
| AWS Group | Ideal Clearance |
|---|---|
| BAlSi | – 0.000-0.002″ (0.000-0.051mm) for furnace brazing in a vacuum atmosphere and clad brazing sheet in a salt bath – 0.002-0.008″ (0.051-0.203mm) for lap length less than 0.250″ (6.35mm) – 0.008-0.010″ (0.203-0.254mm) for lap length greater than 0.250″ (6.35mm) |
| BCuP | – 0.001-0.005″ (0.025-0.127mm) no flux and for flux brazing of joint length less than 1″ (25.4mm) |
| BAg | – 0.002-0.005″ (0.051-0.127mm) flux brazing – 0.000-0.002″ (0.000-0.051mm) atmosphere brazing |
| BCu | – 0.000-0.002″ (0.000-0.051mm) atmosphere brazing |
| BCuZn | – 0.002-0.005″ (0.051-0.127mm) flux brazing |
2: Clean Before You Braze
Contaminants on the metal surface can inhibit the capillary process for the filler metal. This will reduce the strength of the joint being brazed. Chemical and mechanical cleaning processes can be used.
Chemical
- petroleum spray
- chlorinated solvents
- vapor de-greasing
- emulsion spray
- alkaline soak
- acid pickling
- trichlor solvents
Mechanical
- sandblasting (exercise caution to avoid damage to the metal surface)
- machining
- grinding
- brushing (wire brush)
3: Setting Up Joints To Be Brazed
Make the minimally necessary use of vises or clamps needed to enable the joint to be self-supporting (if possible). Avoid or at least be aware of distortions caused by heat-absorbing material such as the clamps.
4: Flux Does Not Eliminate the Need to Clean
Flux helps with oxidation problems caused by oxygen in the air and gas. Too much oxidation will interfere with the capillary effect of the filler alloy. Some filler metals already contain agents that act as flux.
5: Brazing
The brazing method selected depends on the type and size of the job to be performed. For smaller jobs, oxy-acetylene torch brazing is a common approach. In other types of jobs, the brazing processes such as resistance, induction, vacuum and atmosphere furnace brazing can be more efficient. One tip when brazing is to realize that filler metals attract to the highest temperature surface. Therefore if you heat directly on the joint surface the brazing alloy may not fill the joint. Instead, the goal is to heat the interior facing surfaces to the correct temperature, and by situating the brazing alloy close to the joint to be brazed.
6: Cleaning After Brazing
Residues can often be removed with a hot water bath after the filler has solidified. If this doesn’t crack the residue off, try a water jet and wire brush. As a last resort a mild acid bath. Follow the manufacturer’s directions in order to avoid acid etching on the brazed metal.
7: Consider A Multi-Purpose Brazing Rod
Simplify the types of brazing rods needed by using a versatile product such as the HTS-2000. It works on all non-ferrous alloys including “all” aluminum alloys (even the ones that cannot be welded), magnesium aluminum mixtures, zinc, die cast, pot metal, copper, bronze, Nickel, Titanium and galvanized parts.

Video Demonstration
Processes
Generally, brazing processes are specified according to heating methods (sources) of industrial significance. Whatever the process used, the filler metal has a melting point above 840ºF (450ºC) but below the base metal and distributed in the joint by capillary attraction.
The brazing processes are:
- Torch brazing
- Furnace brazing
- Induction brazing
- Resistance brazing
- Dip brazing
- Infrared brazing
- Blanket
- Exothermic
Torch Brazing
Torch brazing is commonly used for smaller production runs or one assembly. This type of brazing is performed by heating with a gas torch set to the proper required composition, and an appropriate flux. This depends on the temperature and heat amount required. The fuel gas (acetylene, propane, city gas, etc.) may be burned with air, compressed air, or oxygen.
Brazing filler metal may be pre-placed at the joint in the forms of rings, washers, strips, slugs, or powder, or it may be fed from hand-held filler metal in wire or rod form. In any case, proper cleaning and fluxing are essential.
For manual torch brazing, the torch may be equipped with a single tip, either single or multiple flame. Manual torch brazing is particularly useful on assemblies involving sections of unequal mass. Welding machine operations can be set up where the production rate allows, using one or several torches equipped with single or multiple flame tips. The machine may be designed to move either the work or torches, or both. For premixed city gas-air flames, a refractory type burner is used.
Furnace Brazing
Furnace brazing is used extensively where the parts to be brazed can be assembled with the brazing filler metal in form of wire, foil, filings, slugs, powder, paste, or tape is pre-placed near or in the joint. This process is particularly applicable for high production brazing. Fluxing is employed except when an atmosphere is specifically introduced in the furnace to perform the same function. Most of the high production brazing is done in a reducing gas atmosphere, such as hydrogen and combusted gases that are either exothermic (formed with heat evolution) or endothermic (formed with heat absorption). Pure inert gases, such as argon or helium, are used to obtain special atmospheric properties.
A large volume of furnace brazing is performed in a vacuum, which prevents oxidation and often eliminates the need for flux. Vacuum brazing is widely used in the aerospace and nuclear fields, where reactive metals are joined or where entrapped fluxes would be intolerable. If the vacuum is maintained by continuous pumping, it will remove volatile constituents liberated during brazing. There are several base metals and filler metals that should not be brazed in a vacuum because low boiling point or high vapor pressure constituents may be lost. The types of furnaces generally used are either batch or contiguous. These furnaces are usually heated by electrical resistance elements, gas or oil, and should have automatic time and temperature controls. Cooling is sometimes accomplished by cooling chambers, which either are placed over the hot retort or are an integral part of the furnace design. Forced atmosphere injection is another method of cooling. Parts may be placed in the furnace singly, in batches, or on a continuous conveyor.
A vacuum is a relatively economical method of providing an accurately controlled brazing atmosphere. Vacuum provides the surface cleanliness needed for good wetting and flow of filler metals without the use of fluxes. Base metals containing chromium and silicon can be easily vacuum brazed where a very pure, low dew point atmosphere gas would otherwise be required.
Induction Brazing
In this process, the heat necessary to braze metals is obtained from a high frequency electric current consisting of a motor-generator, resonant spark gap, and vacuum tube oscillator. It is induced or produced without magnetic or electric contact in the parts (metals). The parts are placed in or near a water-cooled coil carrying alternating current. They do not form any part of the electrical circuit. The brazing filler metal normally is pre-placed.
Careful design of the joint and the coil setup are necessary to assure that the surfaces of all members of the joint reach the brazing temperature at the same time. Flux is employed except when an atmosphere is specifically introduced to perform the same function.
The equipment consists of tongs or clamps with the electrodes attached at the end of each arm. The tongs should preferably be water-cooled to avoid overheating. The arms are current carrying conductors attached by leads to a transformer. Direct current may be used but is comparatively expensive. Resistance welding machines are also used. The electrodes may be carbon, graphite, refractory metals, or copper alloys according to the required conductivity.
Resistance
The heat necessary for resistance brazing is obtained from the resistance to the flow of an electric current through the electrodes and the joint to be brazed. The parts comprising the joint form a part of the electric circuit. The brazing filler metal, in some convenient form, is pre-placed or face fed. Fluxing is done with due attention to the conductivity of the fluxes. (Most fluxes are insulators when dry.) Flux is employed except when an atmosphere is specifically introduced to perform the same function. The parts to be brazed are held between two electrodes, and proper pressure and current are applied. The pressure should be maintained until the joint has solidified. In some cases, both electrodes may be located on the same side of the joint with a suitable backing to maintain the required pressure.
Dip Brazing
There are two methods of dip brazing:
- chemical bath dip brazing
- molten metal bath dip brazing.
Chemical Bath Dip Brazing
In chemical bath dip brazing, the brazing filler metal, in suitable form, is pre-placed and the assembly is immersed in a bath of molten salt. The salt bath furnishes the heat necessary for brazing and usually provides the necessary protection from oxidation; if not, a suitable flux should be used. The salt bath is contained in a metal or other suitable pot, also called the furnace, which is heated from the outside through the wall of the pot, by means of electrical resistance units placed in the bath, or by the I2R loss in the bath itself.
Molten Metal Bath Dip Brazing
In molten metal bath dip brazing, the parts are immersed in a bath of molten brazing filler metal contained in a suitable pot. The parts must be cleaned and fluxed if necessary. A cover of flux should be maintained over the molten bath to protect it from oxidation. This method is largely confined to brazing small parts, such as wires or narrow strips of metal. The ends of the wires or parts must be held firmly together when they are removed from the bath until the brazing filler metal has fully solidified.
Infrared Brazing
Infrared heat is radiant heat obtained below the red rays in the spectrum. While with every “black” source there is sane visible light, the principal heating is done by the invisible radiation. Heat sources (lamps) capable of delivering up to 5000 watts of radiant energy are commercially available. The lamps do not necessarily need to follow the contour of the part to be heated even though the heat input varies inversely as the square of the distance from the source. Reflectors are used to concentrate the heat.
Assemblies to be brazed are supported in a position that enables the energy to impinge on the part. In some applications, only the assembly itself is enclosed. There are, however, applications where the assembly and the lamps are placed in a bell jar or retort that can be evacuated, or in which an inert gas atmosphere can be maintained. The assembly is then heated to a controlled temperature, as indicated by thermocouples. The part is moved to the cooling platens after brazing.
Blanket Brazing
Blanket brazing is another of the processes used for brazing. A blanket is resistance heated, and most of the heat is transferred to the parts by two methods, conduction and radiation, the latter being responsible for the majority of the heat transfer.
Exothermic Brazing
Exothermic brazing is another special process by which the heat required to melt and flow a commercial filler metal is generated by a solid state exothermic chemical reaction. An exothermic chemical reaction is defined as any reaction between two or more reactants in which heat is given off due to the free energy of the system. Nature has provided us with countless numbers of these reactions; however, only the solid state or nearly solid state metal-metal oxide reactions are suitable for use in exothermic brazing units. Exothermic brazing utilizes simplified tooling and equipment.
The process employs the reaction heat in bringing adjoining or nearby metal interfaces to a temperature where pre-placed brazing filler metal will melt and wet the metal interface surfaces. The brazing filler metal can be a commercially available one having suitable melting and flow temperatures. The only limitations may be the thickness of the metal that must be heated through and the effects of this heat, or any previous heat treatment, on the metal properties.
The Use of Channel Flux to Reduce Manufacturing Cost and Improve Throughput. It is Available With Aluminum Based and Zinc Based Alloys.
Selection of Base Brazing Metals
In addition to the normal mechanical requirements of the base metal in the brazement, the effect of the brazing cycle on the base metal and the final joint strength must be considered. Cold-work strengthened base metals will be annealed when the brazing process temperature and time are in the annealing range of the base metal being processed. “Hot-cold worked” heat resistant base metals can also be brazed; however, only the annealed physical properties will be available in the brazement. The brazing cycle will usually anneal the cold worked base metal unless the brazing temperature is very low and the time at heat is very short. It is not practical to cold work the base metal after the brazing operation.
When a brazement must have strength above the annealed properties of the base metal after the brazing operation, a heat treatable base metal should be selected. The base metal can be an oil quench type, an air quench type that can be brazed and hardened in the same or separate operation, or a precipitation hardening type in which the brazing cycle and solution treatment cycle may be combined. Hardened parts may be brazed with a low temperature filler metal using short times at temperature to maintain the mechanical properties.
Ease of Brazement
The strength of the base metal has an effect on the strength of the brazed joint. Some base metals are also easier to braze than others, particularly by specific brazing processes. For example, a nickel base metal containing high titanium or aluminum additions will present special problems in furnace brazing. Nickel plating is sometimes used as a barrier coating to prevent the oxidation of the titanium or aluminum, and it presents a readily wettable surface to the brazing filler metal.

Brazing Filler Metals
For satisfactory use in brazing applications, brazing filler metals must possess the following properties:
- The ability to form brazed joints possessing suitable mechanical and physical properties for the intended service application.
- A melting point or melting range compatible with the base metals being joined and sufficient fluidity at brazing temperature to flow and distribute into properly prepared joints by capillary action.
- A composition of sufficient homogeneity and stability to minimize separation of constituents (liquation) under the brazing conditions to be encountered.
- The ability to wet the surfaces of the base metals being joined and form a strong, sound bond.
- Depending on the requirements, the ability to produce or avoid base metal-filler metal interactions.
Brazing Gray Cast Iron
Gray cast iron can be brazed with very little or no preheating.
For this reason, broken castings that would otherwise need to be dismantled and preheated can be brazed in place.
A nonferrous filler metal such as naval brass (60 percent copper, 39.25 percent zinc, 0.75 percent tin) is satisfactory for this purpose. This melting point of the nonferrous filler metal is several hundred degrees lower than the cast iron; consequently, the work can be accomplished with a lower heat input, the deposition of metal is greater and the brazing can be accomplished faster. Because of the lower heat required for brazing, the thermal stresses developed are less severe and stress relief heat treatment is usually not required.
The preparation of large castings for brazing is much like that required for welding with cast iron rods.
The joint to be brazed must be clean and the part must be sufficiently warm to prevent chilling of filler metal before sufficient penetration and bonding are obtained.
When possible, the joint should be brazed from both sides to ensure uniform strength throughout the weld. In heavy sections, the edges should be beveled to form a 60 to 90 degree V.
Brazing Malleable Iron
Malleable iron castings are usually repaired by brazing because the heat required for fusion welding will destroy the properties of malleable iron. Because of the special heat treatment required to develop malleability, it is impossible to completely restore these properties by simply annealing. Where special heat treatment can be performed, welding with a cast iron filler rod and remalleabilizing are feasible.